Pharmaceutical LIMS & Stability Study Management
Managing QC testing in the highly regulated pharmaceutical manufacturing and tracking raw ingredients to final product has never been so easy.
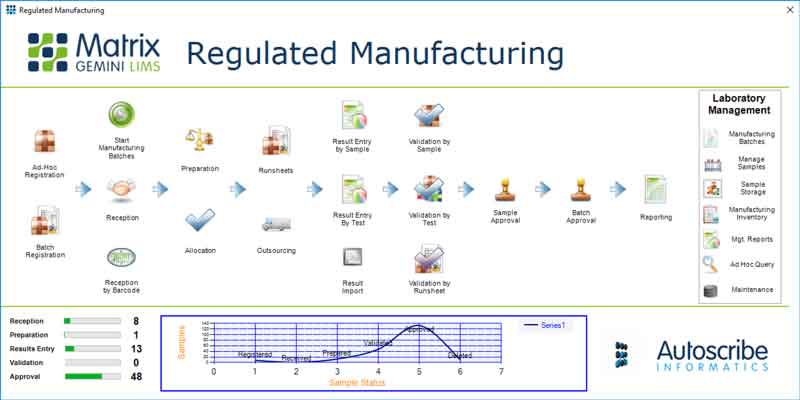
Pharmaceutical LIMS Defined by Standards
Designed specifically for regulated manufacturing industries such as pharmaceutical manufacturing, Xybion Pharmaceutical LIMS is ideal for highly regulated industries working to standards such as FDA 21 CFR Part 11, ISO 17025 and cGxP.
Developed according to our ISO 9001 accredited quality system the solution is supported by comprehensive validation documentation providing a head start to validation activities. Unique graphical configuration tools allow the Pharmaceutical LIMS to be adapted to your specific needs while ensuring that the validated core software remains untouched.
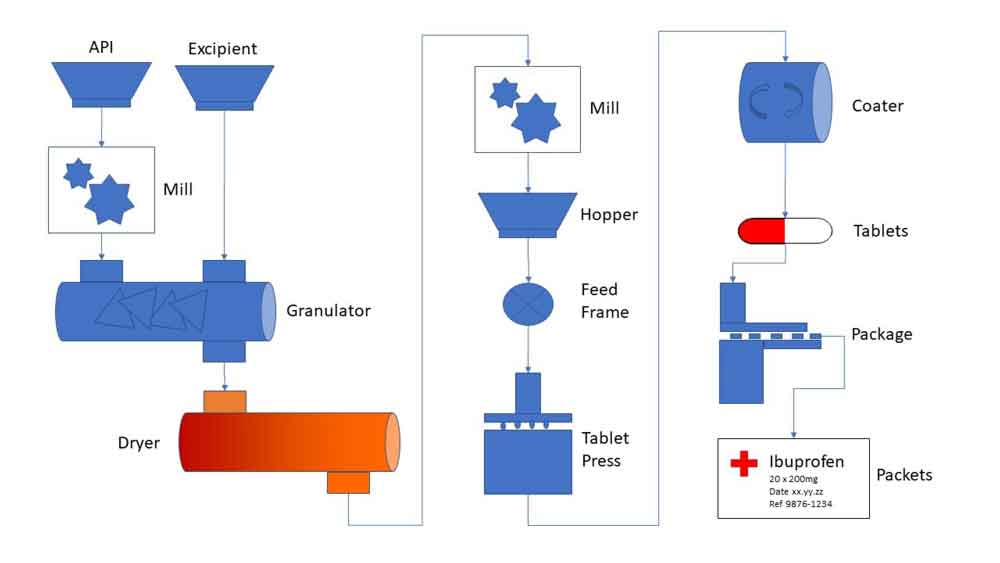
Raw Ingredients to Final Product Tracking
Whether testing finished goods with the creation of a final Certificate of Analysis (CofA), testing incoming raw materials, or testing intermediate product, Xybion Pharmaceutical LIMS helps you automate and control your sample testing and record data results to conform to regulatory requirements.
The solution drives quality throughout the pharmaceutical manufacturing flow by managing and tracking the samples taken and ensuring they can be quickly related to the relevant product and batch.

Stability Study Testing
Managing pharmaceutical stability testing can be very demanding, especially with small to medium size companies developing and producing over the counter, generic and new prescription products. Some companies outsource the actual inventory management and testing requirements, but they are still required to track progress and report results as part of their QA or Development process.
Manually ensuring that approved protocols are followed precisely, including “pulls” being made on schedule with the appropriate tests being performed, can be both time consuming and tedious.
The laboratory must also ensure that any laboratory informatics system can interface with other enterprise system such as Enterprise Resource Planning systems (ERP) or Manufacturing Resource Planning Systems (MRP), etc. Enabling the laboratory to add value to the manufacturing process is key.




