Agriya Analitika latest news
Advantages of a LIMS
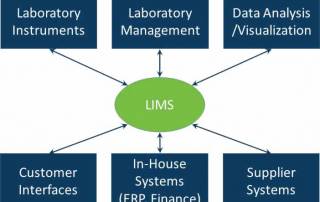
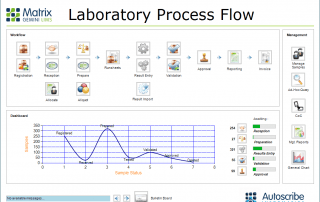
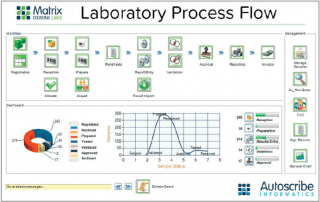
LIMS have evolved to manage the laboratory workflow, organize the data efficiently and support the business of the laboratory. In addition they can drive and support regulatory and standards compliance.