Determining the pros and cons of operating an in-house laboratory or sending samples out to an external laboratory to be tested is increasingly topical. Whether your products are toys or fish pies, cars or shoes, blood products or water, nearly all testing can be performed by a contract laboratory.
Reasons for Sample Tracking
However, if you do decide to outsource laboratory testing there is still a real need for sample tracking within your organization. The following are the key reasons why the use of a sample tracking application is so important:
- Each sample can be uniquely identified by using an automatic number generator in the Sample Tracking application. Labels for each sample can be automatically printed to include bar-codes as well as text and affixed to the samples prior to dispatch. In addition, a worklist can be generated as a report from the Sample Tracking application to provide a contract laboratory with clear instructions.
- The target dates for return of results can be included in the instructions. These target dates can be specified individually for each sample or can be generated automatically if a default time is used. This default time can be set for each test individually, if needed, e.g. a microbiology test will require more time than a pH test.
- The Sample Tracking application can track samples sent to different contract laboratories. This is a common need as different laboratories may have specialist expertise and equipment. Comments about the capabilities and performance of each laboratory can also be stored alongside contact details.
- The cost of testing can also be held within the Sample Tracking application. This allows the creation of management reports that record the number of samples, and associated costs, for each contract laboratory over a selected period of time.
- On completion of testing the test results from the contract laboratories can be uploaded electronically to the Sample Tracking application. At this point in-house specification limits can be applied to the results, thus giving quick indications of problems that might exist.
- Trend analysis of the test results can be offered as an option for the Sample Tracking application to further compare samples, batches or projects to spot emerging trends.
- In the case of suspect results a CAPA module could be provided as an option with the Sample tracking application.
Overall, the Sample Tracking application provides full sample tracking capability, complete with data analysis. Depending on the size of the organization the need may only be for a single user system, although multiple user systems would also be available.
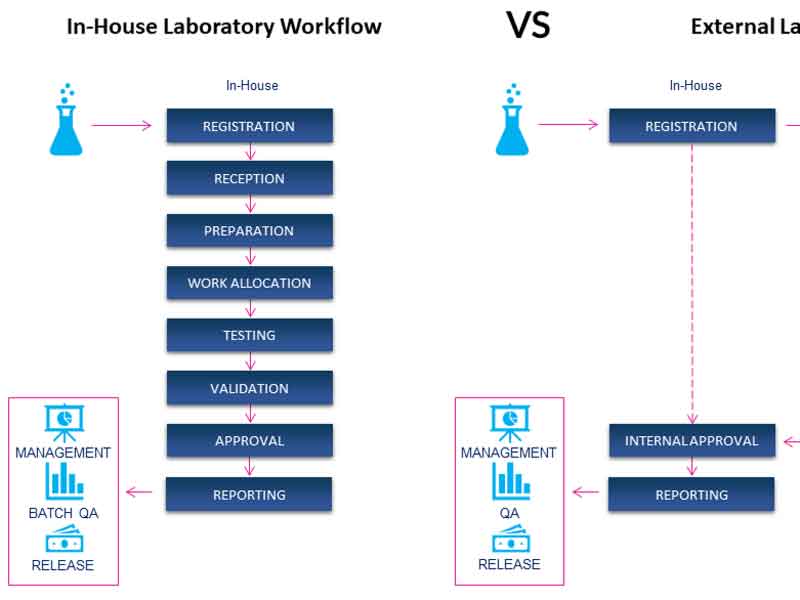
In-House Vs External Laboratory
An in-house laboratory will perform it’s own testing and approve the quality of the product for release themselves.

When using an external laboratory a variety of samples will be sent to the external laboratory for testing. In order to track those samples the internal laboratory will need to register the samples and provide a barcode so that results from the external laboratory can be referenced back to the correct sample. Test result data as well as a certificate of analysis may be passed back from the external laboratory, and these analytical results can be stored in a Sample Tracking application.
This allows the in-house staff to check the results and to perform data analysis across samples and/or projects and to derive other information for management purposes. A Sample Tracking application enables the in-house team to import the data coming from the external laboratory, keep that data in one database for ease of management, and to perform these reporting functions.
The trade-off between running an in-house laboratory vs outsourcing the laboratory testing is one of resources, cost, speed, and confidence.
Resources
All too often the internal laboratory is seen as a cost center. It requires expensive specialist staff, highly priced laboratory equipment and costly laboratory consumables. Using a contract laboratory can remove these overheads, though the in-house technical/project manager will still need to assess results to be sure product standards are being maintained.

Speed
With an in-house laboratory turn-around time should, arguably, be faster and prioritizing urgent requests should be simpler. An external laboratory will always have some latency simply because of the time it takes to ship samples and because of competing requests from other customers, although this risk can be mitigated by ensuring Service Level Agreements (SLAs) are in place. The trade-off can often come down to how important speed of response is to product release, for example, but may also be down to availability of analytical skills and required instrumentation.
Confidence
Potentially organizations may lack confidence in the service provided by contract laboratories, and here again the implementation of enforceable SLAs can help. Organizations should, however, be looking at contract testing organizations that meet recognized standards. The scope of ISO 17025 ‘General requirements for the competence of testing and calibration laboratories’ states; Laboratory customers, regulatory authorities, organizations and schemes using peer-assessment, accreditation bodies, and others use this document (i.e. ISO 17025) in confirming or recognizing the competence of laboratories.
A New Pressure on Internal Laboratories
During the current Covid-19 pandemic organizations have been looking at ways to reduce the number of staff on site. While innovative staff rosters and schedules have been introduced, some organizations may have furloughed some, or all, of their analytical staff in favor of outsourced testing. If this is proven to be successful it will further increase the pressure on internal laboratories.
Driving Business Efficiency With a LIMS
No matter by who or where the testing is done one of the keys to making the analytical laboratory work is the management of data. While it is possible for third party laboratories to send results and certificates of analysis in the form of files by email to their customers, this can risk inboxes becoming overfilled and vital results being overlooked.
The alternative methodology is to provide the information in a format that a Sample Tracking system can upload directly, and associate it with the relevant samples, product, or batch. This information is available more quickly as it does not need to be processed manually, and it also avoids typographical errors through manual data entry. If certificates of analysis are provided these can also be associated with the relevant samples, product, or batch. Adopting this approach ensures that the data needed to make product usage or release decisions is quickly available, accurate and accessible.
An additional benefit is that the data is available in the Sample Tracking application and can be used for data analytics. The use of data analytics provides further opportunities to investigate trends and outliers, optimize processes, and make further cost savings. Statistical Process Control charts enable sample results within and across batches to be compared to find trends and warn of possible issues before they cause a line stop, or worse.
Management charts can be derived from the imported result data showing efficiency of the contract laboratory through turn-around time charts and other data. Multiple data sets from across the organization, including the Sample Tracking application, can be interrogated by data analytics tools, such as Microsoft’s PowerBI, to provide even more granularity and flexibility during investigative research.
Summary
It is a mistake to think that outsourcing your laboratory means you do not need a system for tracking samples and the resulting data. We can call this a LIMS even when an internal laboratory does not exist. The heart of every business, the gold dust that enables innovation, is increasingly seen as the data it creates. A LIMS is specifically designed to manage and retain key analytical information, from wherever it comes.
The true benefit of a LIMS is having that data at your fingertips for every batch of every day, together with the ability to access that data in multiple ways. This data provides a defendable QC framework for the business, if challenged, and ensures that you can benefit from intelligent use of the data to increase efficiency and profitability. Quite apart from anything else a LIMS removes the nightmare of an in-tray full of unfiled Certificates of Analysis from an external laboratory and instead records them correctly and automatically against the relevant sample and batch codes for instant recall.
A LIMS is therefore an invaluable, indeed essential, business management tool whether your laboratory is in-house or external.
An additional benefit is that the data is available in the Sample Tracking application and can be used for data analytics. The use of data analytics provides further opportunities to investigate trends and outliers, optimize processes, and make further cost savings. Statistical Process Control charts enable sample results within and across batches to be compared to find trends and warn of possible issues before they cause a line stop, or worse.
Management charts can be derived from the imported result data showing efficiency of the contract laboratory through turn-around time charts and other data. Multiple data sets from across the organization, including the Sample Tracking application, can be interrogated by data analytics tools, such as Microsoft’s PowerBI, to provide even more granularity and flexibility during investigative research.
How to Buy a LIMS – The Definitive Guide
The LIMS Selection Guide aims to help anyone considering the purchase of a new or replacement of Laboratory Management System.