A recent theme in a number of recent FDA warning letters has been analytical data integrity. This is more, however, than simply connecting instruments to laboratory software for automatic data capture. Recent warning letters have cited the need to ensure records can only be accessed by relevant personnel, that audit trails are complete, and that all records can be accounted for.
Clearly the FDA are taking the ALCOA and ALCOA+ data integrity principles to heart. This article shows how a Laboratory Information Management System (LIMS) can help address some of the concerns raised by the FDA and other regulatory agencies.
Control Access to Data
Two recent warning letters, Company A and Company B, were cited for “failing to exercise appropriate controls over computer or related systems to assure that only authorized personnel institute changes in master production and control records, or other records”. Although records were kept electronically, they could be altered or deleted without due control.
It is standard practice within a laboratory to allow only appropriate users the ability to access and alter records, and to maintain a complete audit trail of those changes. Ensuring the integrity of electronic test data is a key point during FDA audits, and to maintain compliance laboratories should ensure that:
- Data is captured electronically into the LIMS from laboratory instruments, with no manual intervention,
- All relevant raw data and metadata is recorded, in addition to any data derived from it.
- Each LIMS user has their own username and password, and there are no shared accounts.
- Users only have access to the data they need to perform their role.
- Key checkpoints, such as data validation and approval, enforce the use of passwords, and where appropriate electronic signatures.
- If a data record is over-written a record of who made the changes and why must be kept for audit purposes.
These rules apply not only to the LIMS in your lab but also to other laboratory informatics systems such as chromatography data systems (CDS) which may be integrated with the LIMS. The aim is a defendable audit trail demonstrating clear control of the chain of data throughout its life, from data capture to reporting, ensuring data integrity.
Increase Instrument Integration
Three warning letters, Company B, Company C, and Company D, asked companies to consider “Technological improvements to increase the integration of data generated through electronic systems from standalone equipment (e.g., balances, pH meters, water content testing) into the LIMS network”. This wording has started to appear in FDA warning letters more frequently and is an indication that integrating instruments into the LIMS, wherever feasible, is now the norm.
Autoscribe has solutions to integrate different types of instruments which broadly fall into three categories:
- Serial Interfaces
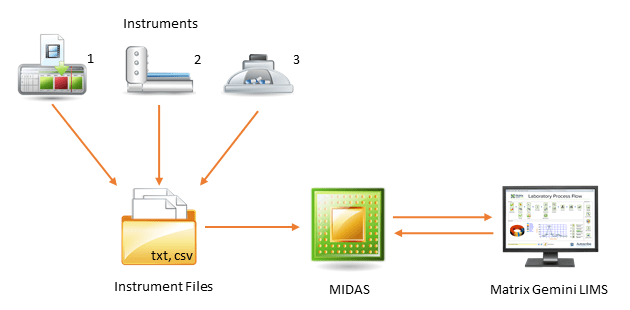
Simple instruments such as balances and pH meters often have a serial/USB interface that may be used to capture readings from the instrument. Matrix Gemini LIMS has a built-in serial interface to connect these instruments. - File Interfaces
More complex instruments can often output a file that can be directly read by the LIMS. These files are typically .txt or .csv format. The Matrix Gemini LIMS MIDAS module will automatically read these files, extract the required analytical and meta data, and pass it to the LIMS. As the interface format is so general it suits many unidirectional instrument interfaces whoever the manufacturer. Matrix Gemini LIMS can also create and pass back formatted files to the instruments if bidirectional interfaces are required. - API Interfaces
For more complex interfaces, such as interacting with corporate SAP or MRP (Manufacturing Resource Planning) systems, and with complex instruments such as Clinical Analyzers a direct bi-directional link between the two systems is required. Systems typically have an Application Programming Interface (API) which allows data to be sent backwards and forwards between the systems.
Benefits of Direct Instrument integration to a LIMS
Whether instruments and systems are connected via serial interface, file transfer, API or through some other means such as an intermediary Instrument Management System (IMS) or middleware the benefits are similar.
When connecting systems and instruments directly to your LIMS you can:
- Ensure the correct instrument is used and prevent result approval if not.
- Check the instrument is qualified and calibrated and block result entry if not.
- Calculate final results from imported raw data within the LIMS.
- Approve and electronically sign the results within the LIMS.
- Prevent transcription errors.
Summary
Quality and regulatory departments within companies running laboratories must be aware of the need to manage electronic data, who can access it, and how it is used throughout the lifetime of the data. Data integrity must be built into the systems.
An adverse finding from an FDA audit, or being unable to track and trace analytical result data for historical batches subject to adverse reaction reports or customer complaints, is not the time to discover data integrity issues.