LIMS Ensures FDA 21 CFR Part 11 Compliance
LIMS helps laboratories comply to FDA 21 CFR Part 11 by enforcing signature policies for all electronic records. Ensuring electronic records and electronic signatures can be relied upon in the same way as paper records and handwritten signatures.
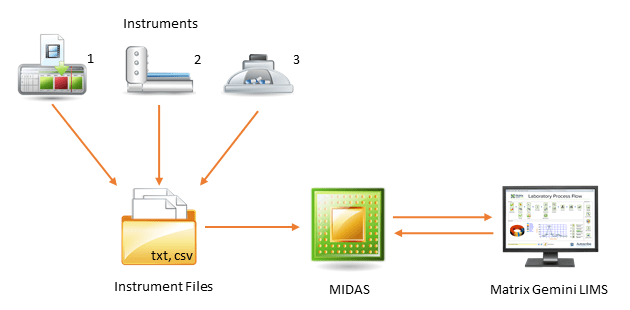
Simplifying Results Collection and Approval in Laboratories
Direct instrument integration into the LIMS eliminates this possibility and helps meet data integrity and validity requirements.
Advantages of a LIMS
LIMS have evolved to manage the laboratory workflow, organize the data efficiently and support the business of the laboratory. In addition they can drive and support regulatory and standards compliance.
Stability Study with Matrix Gemini LIMS
Stability studies are used to assess the shelf life of a product by storing samples under controlled environmental conditions for defined lengths of time prior to testing.
Webinar: Ensuring ISO 17025:2017 compliance in your Laboratory
A cornerstone of the ISO 17025 requirement for laboratories to demonstrate they operate competently and are able to generate valid results.
LIMS Limitation?
LIMS are well known for their ability to manage sample data and their associated test results. However, many lab managers may be surprised at how LIMS can also help with many other laboratory management functions to improve efficiency and help support the needs of industry standards and regulation such as ISO/IEC 17025 and GxP.
LIMS Solutions for the Pharmaceutical Industry
The global pharmaceuticals market is worth over $750 billion a year, a figure that is expected to continue to rise. In a highly competitive and increasingly expensive market place it is important that cost efficiencies are maximized, while meeting all critical compliance requirements.