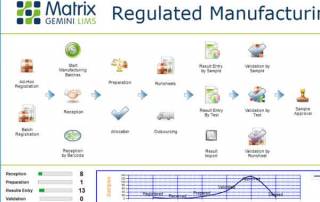
Webinar: Flexible Scope of LIMS for Manufacturing Operations
This webinar provides an insight into how LIMS continues to evolve as the glue that links the disparate elements of manufacturing data and information together to ensure safe and efficient product release.