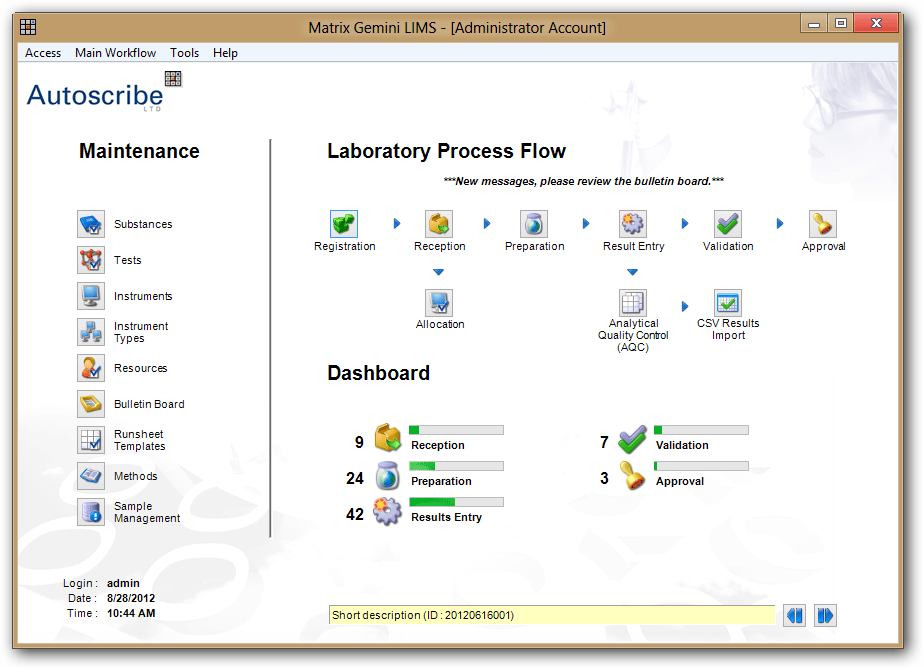
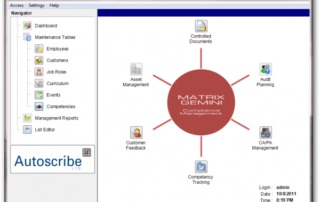
ISO 17025 Documentation within Matrix Gemini LIMS – Corrective & Preventative Actions
ISO/IEC 17025 specifies the general requirements for the competence of a laboratory to carry out tests and/or calibrations, including sampling. In defining competence, the standard requires that there are procedures for the resolution of complaints from customers or others, control of nonconforming testing and/or calibration work and for the implementation and monitoring of any corrective or preventative actions required.